Zynerba Pharmaceuticals, Inc. General Corporate Statement (Form8) - Zynerba Pharmaceuticals (NASDAQ:ZYNE) - Benzinga

Zynerba Pharmaceuticals - Whether They've Cracked The Code For Fully Methylated FX Syndrome Patients (NASDAQ:ZYNE) | Seeking Alpha

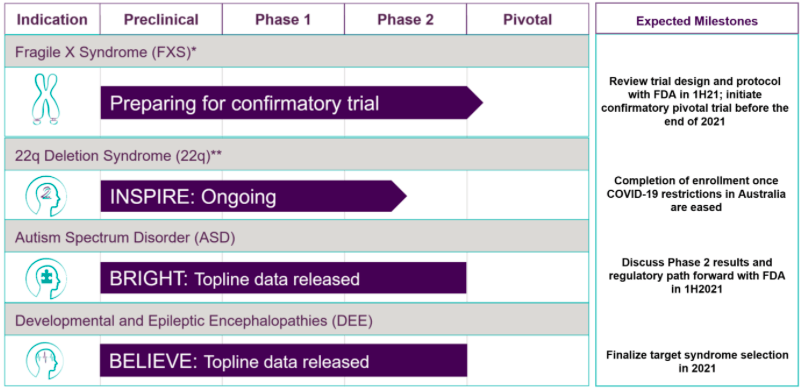
Zynerba Pharmaceuticals Inc receives guidance from the US Food and Drug Administration on a confirmatory Phase 3 trial of Zygel in patients with Fragile X syndrome

Zynerba Pharmaceuticals Inc's CBD skin gel Zygel offers a potential goldmine of therapies for Fragile X syndrome, and other conditions







/arc-anglerfish-arc2-prod-pmn.s3.amazonaws.com/public/YST6UFBNEVBAXNOUKDYSHHF4AM.PNG)